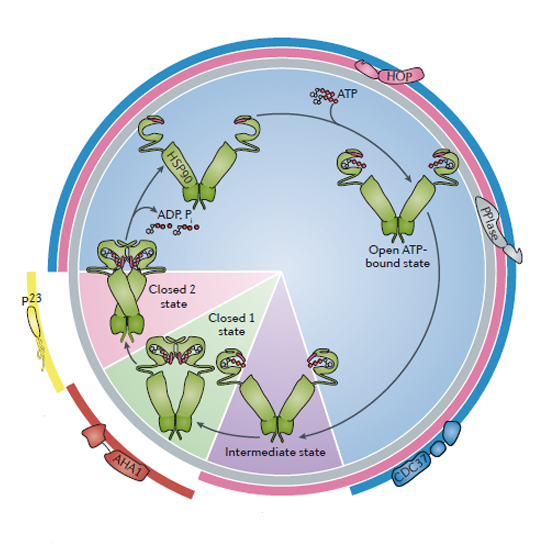
The bound co‐chaperone contacts the three main switch segments – the lid (magenta), catalytic loop (green) and N‐terminal helix/strand (blue) but only in their ATP‐bound ‘tense’ conformations. D: Close up of P23/Sba1 complex with HSP90. C: P23/Sba1 binds across the interface between the dimerized N‐terminal domains in the ATP‐bound closed state. A single molecule of CDC37 and of the kinase bind asymmetrically to an HSP90 dimer. B: Negative stain electron microscope single particle reconstruction of an HSP90‐CDC37‐CDK4 client protein complex. A: The middle and C‐terminal regions of CDC37 bind to the lid segment in the N‐domain of HSP90 and prevent its closure on ATP‐binding. Biopolymers Published by Wiley Periodicals, Inc.ĪTPase‐regulatory co‐chaperones. Biopolymers 105: 594-607, 2016.ĪTP client protein cochaperone conformational change molecular chaperones. Despite this, the actual function that HSP90s conformationally-coupled ATPase activity provides in its biological role as a molecular chaperone remains obscure. Pharmacological inhibition of HSP90 ATPase activity results in degradation of client proteins in vivo, and is a promising target for development of new cancer therapeutics. Work over the last two decades has revealed a conformational cycle critical to the biological function of HSP90, coupled to an inherent ATPase activity that is regulated and manipulated by many of the co-chaperones proteins with which it collaborates. The HSP90 molecular chaperone is involved in the activation and cellular stabilization of a range of 'client' proteins, of which oncogenic protein kinases and nuclear steroid hormone receptors are of particular biomedical significance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
